“Process intensification” is one of those big umbrella terms that unfairly implies a concept that comes with an instruction manual. It doesn’t. It isn’t prescriptive. It doesn’t have to start in a specific place, and it really never ends.
- A Deeper Look At What's Driving Dependence On Biotech Contract Manufacturers
- Developing A Process Performance Qualification Master Plan For Gene Therapies
- What Time Is Best To Bring My CDMO On Board?
- Single Use In Biopharma: Beyond Savings & Sustainability
- Unpacking The Importance Of Lipid Nanoparticle Production Platforms
- Cell Counting Is Growing More Important Amid Advanced Therapy’s Rise
- FDA Publishes Final Guidance On Reporting Listed Drug Amounts
EDITOR'S DESK
-
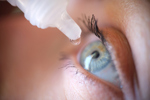
Eying Up The Era Of Topical Biologics
Claris Bio’s phase 1/2 clinical trial in patients with Stage 2 or 3 Neurotrophic Keratitis (NK) is breaking new ground in the development of topically administered biologic therapies.
The biopharmaceutical industry is building manufacturing capacity at breakneck speed. According to PhRMA, there are 1,580-and-counting biopharma manufacturing facilities in the U.S. alone. Problem is, we're way short on filling those facilities with skilled labor. NIIMBL wants to change that. Here's how, and a great opportunity for big pharma, biotech sponsors, CDMOs, academics, and more to help.
The career of Debra Weiss, RN, MSN, and COO at the biopharma Gates Medical Research Institute (Gates MRI), offers a case study in creating success through service-oriented leadership.
Check out the results of DeciBio's Antibody-Drug Conjugates 2024 Industry Survey, which predict another bullish year for the growing modality.
The U.S. imported $6.95 billion worth of pharmaceuticals from China in 2022. That was 8x the amount we imported in 2021. A proposed Act of Congress puts that supply, and U.S. biotech itself, in serious peril.
GUEST COLUMNISTS
-
Developing A Process Performance Qualification Master Plan For Gene Therapies
This article provides considerations and a framework for a Process Performance Qualification Master Plan in the gene therapy industry. It is useful for sponsors with any level of experience.
-
What Time Is Best To Bring My CDMO On Board?
The answer might be sooner than you think and entail more transparency than you're used to.
-
Unpacking The Importance Of Lipid Nanoparticle Production Platforms
Batch processing platforms for LNPs lack efficiency, appropriate controls, and yield optimization. Continuous manufacturing platforms may address some of those issues.
-
Cell Counting Is Growing More Important Amid Advanced Therapy’s Rise
You can't improve what you don't measure. This discussion explores advances in cell counting technology and the growing number of ways it aids the biotech industry and beyond.
-
FDA Publishes Final Guidance On Reporting Listed Drug Amounts
Despite earlier efforts to shore up the U.S. drug supply chain, COVID-19 still brought shortages. A new FDA reporting system aims to reduce future supply risks.
-
2024 CIO Survey Reveals IT Budgets, AI Priorities For Life Sciences Companies
Clarkston Consulting conducted its third annual Chief Information Officer (CIO) Survey to understand IT budgets of life sciences companies, how AI priorities fit into those budgets, and more.
-
AI And Pharmaceutical Development: WHO Calls For Ethical Framework, Good Governance
The World Health Organization (WHO) recently released a document titled Benefits and risks of using artificial intelligence for pharmaceutical development and delivery describing the ethical issues and governance considerations for using AI in drug discovery, preclinical development, and clinical trials.
-
Digitizing CMC Knowledge Management
Systems that enhance know-how retention stretch from utterly simple to enterprise-scale. Here are some ideas for incorporating them into your knowledge management strategy.
BIOPROCESSING WHITE PAPERS
-
Next Gen Plasmid Technology: Improve Performance, Safety, Manufacturing
Nanoplasmids have paved the way for facilitating a wide range of cell and gene therapeutic applications. Gain an overview of a novel technology and specific benefits.
-
From Bench To Billions: Facilitating Scale-Up Of Adherent Cultures For Cell And Gene Therapies6/27/2022
We review what researchers should consider when they are creating a risk-mitigating adherent culture process for cell and gene therapy scale-up.
-
Risk Assessment As A Process Quality Assurance Tool8/12/2022
Discover how an effective approach to risk management can further ensure the delivery of a high-quality drug or medicine to the patient.
-
The Promise Of PCM: Getting To Maturity9/19/2023
PCM offers a number of advantages, including streamlined development and increased flexibility. Review the key drivers for PCM as well as the hurdles that stand in the way of industry-wide adoption.
-
Manufacturing Of Cytotoxic And Non-Cytotoxic Drugs In A Multiproduct Facility2/15/2023
The experience and expertise of the CMO is crucial to avoid the risk of cross-contamination of the drug product, as failures or weaknesses in this process can pose risk to the end-user.
-
A Dual-Dye Ratiometric Photometry System For Multichannel Verification11/13/2023
Explore the technical and scientific application of dual-dye ratiometric photometry in microtiter plates, upon which a multichannel verification system is based.
BIOPROCESSING APP NOTES & CASE STUDIES
- Direct And Rapid Multi-Attribute Monitoring Of Intact Monoclonal Antibodies By icIEF-UV/MS
- Sf9 Cell Suspension Culture In Corning® Insect Cell Serum-Free Medium
- Optimizing The Formulation For A Monoclonal Antibody
- How High-Performance Syringe Filters Extend The Life Of Analytic Instruments And Columns
- Single Nucleus Dispensing
BIOPROCESS ONLINE CONTENT COLLECTIONS

This collection of articles, produced through a partnership between MilliporeSigma and Bioprocess Online, highlights the need for ongoing investment in resilient biomanufacturing capacity and supply chains to ensure the reliable delivery of health technologies like vaccines, even during crisis conditions.
More Content CollectionsFEATURED PRODUCTS AND SERVICES
ON-DEMAND WEBINARS
- Culturing And Characterizing Organoids And Cancer Spheroids
- How To Get Started With Organ-On-A-Chip Technology
- Optimizing Perfusion Parameters For Intensified Production Of CAR-T Cells
- Innovative Preservation Solutions For Cell Therapy
- De-Risking The Aseptic Drug-Filling Process And Optimizing Production
INDUSTRY NEWS
NEWSLETTER ARCHIVE
- 04.24.24 -- Improve Your Process Development To Accommodate Diverse mAbs
- 04.23.24 -- Genetic Stability Simplified. One Method, Multiple Answers.
- 04.23.24 -- Business Of Biotech Newsletter
- 04.23.24 -- Novel Approaches In Chromatography Process Development
- 04.23.24 -- Women In Biotech With Bill & Melinda Gates MRI's Piper Trelstad, Ph.D.